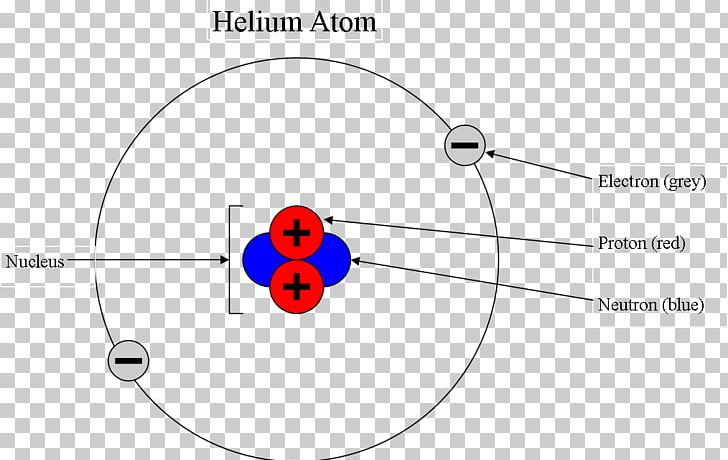
It not only explained the deflection of electron beams on thin metal foils (cf. And fourthly, the structure of atoms with higher atomic number was not given―not even the one of helium―, and likewise the “Aufbau-Principle” of the periodic system of the elements.Ī step forward was made in 1924 by Louis De Broglie in his thesis, assuming a wavy electron motion, and leading to the term “wave mechanics”. Thirdly, Bohr did not deliver a model of a molecule like the H 2-molecule, in contrast to the notification in the title of his article. Secondly, the intrinsic cause for the existence of such exited―meta-stable―energy states could not be found. it was not plausible why the electron does not tumble on the nucleus. However, several questions remained: Firstly, the existence of a minimal ground state (permanent state) could not be explained, i.e. Hence Bohr’s atom model may be considered as the outset of modern quantum mechanics. It is worth mentioning that Planck’s constant h, which he himself denoted as “elementares Wirkungsquantum” (“elemental action quantum”), represents the product of energy and time (delivering the dimension J・s), being equal to the dimension of an angular momentum. Moreover, he found that the angular momenta of the excited states are integral multiples of the angular momentum of the ground state. The essential theorem of his consideration may be expressed by the following statement made in : “In any molecular system consisting of positive nuclei and negative electrons in which the nuclei are at rest relative to each other and the electrons move in circular orbits, the angular momentum of every electron round the centre of its orbit will in the permanent state of the system be equal to h/2π, where h is Planck’s constant.” (Therein, “permanent state” means “ground state” of the electron). His model could widely explain the line-spectrum of hydrogen which had been observed astronomically in certain Space regions as well as in terrestrial experiments using low pressure tubes, and wherein remarkable regularities had been originally found by Balmer, later revealed by Rydberg. 
Starting from Rutherford’s atom model, which assumed a heavy positively charged nucleus in the centre and a light electronic envelope, and applying Planck’s quantum theory of radiation, introduced in 1900 and supplemented by Albert Einstein’s photoelectric effect in 1905, he suggested that―as a result of (UV-) radiative excitation―well-defined orbits were occupied by the electron, being associated to enhanced energy levels. 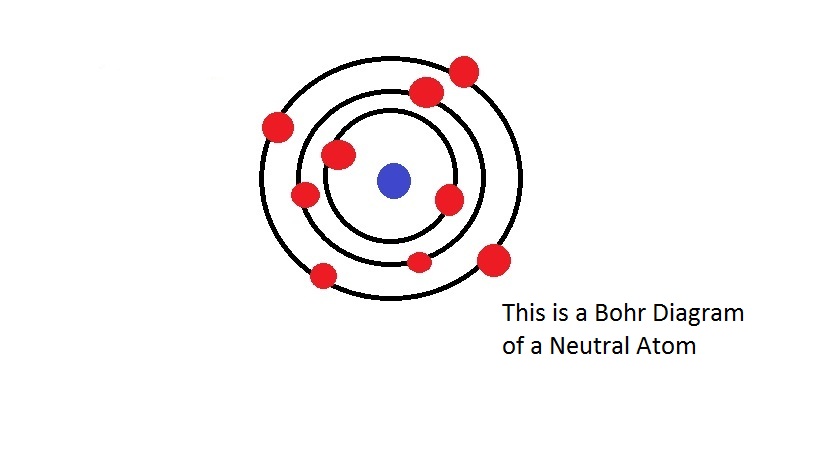
In 1913, the Dane Niels Bohr published an article entitled “On the Constitution of Atoms and Molecules”, focusing on the H-atom. Generally not possible, also not for the H 2 -molecule, and not even Theory which claims that such a-locally and temporally-precise description of any atom or molecule structure is 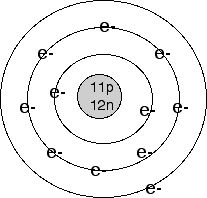
This contradicts the conventional quantum mechanical Limit such a precise description is feasible, being also demonstrated in theĪuthor’s previous work. Marks the limit for precisely describing an atom, whereby at and under this ItĮntails the principal statement that the neon atom does not represent a staticĬonstruction with constant electron distances and velocities, but a pulsatingĭynamic one with permanently changing internal distances. Rough ball-and-stick model can be presented, assuming electron rings instead ofĮlectron clouds, which in the outer shell are orientated as a tetrahedron. Turned out to be too complex for a mathematical modelling. But while this was feasible exactly in the case of helium, the neon atom Treatise, Bohr’s theorem is adapted to the atom models of he lium and of Presumption, a model of the H 2 -molecule could be created whichĮxhibits well-defined electron trajectories, and which enabled computing the In a subsequent treatise, Bohr’s theorem of anĭetermining the ground state of the H-atom, was revealed as an inducement by the -unalterable-electron spin. H-atom could be demonstrated, starting from Bohr’s original model but modifying Mathematical description of electron trajectories in the excited states of the In a previous, primary treatise of the author the
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
